Electronic nose technology in bronchial asthma diagnosis and management
Keywords:
asthma,electronic-nose,volatile organic compoundsAbstract
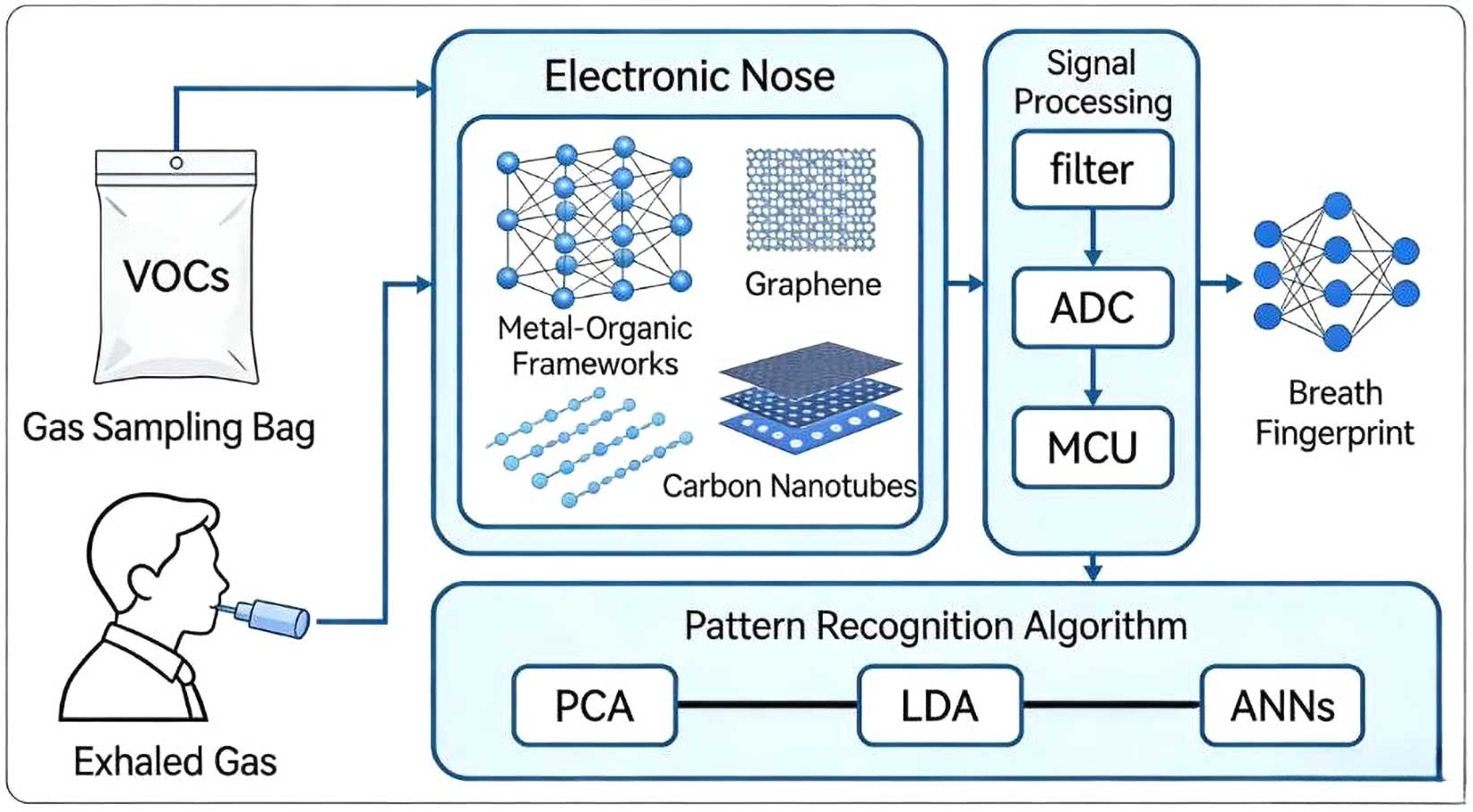
Electronic nose (e-nose) technology, as an emerging non-invasive diagnostic tool, has demonstrated considerable potential in the diagnosis and management of bronchial asthma. By mimicking the human olfactory system, this technology utilizes gas sensor arrays combined with pattern recognition algorithms to enable rapid analysis and identification of volatile organic compounds (VOCs) in the exhaled breath of patients with asthma. Accumulating evidence suggests that e-nose systems can discriminate between asthma patients and healthy individuals with relatively high accuracy, and they have also shown promising performance in asthma phenotyping. Compared with conventional pulmonary function tests, e-nose technology offers distinct advantages, including cost-effectiveness, rapid response, and non-invasiveness. These features make it particularly suitable for populations requiring repeated dynamic assessment or those unable to perform high-quality pulmonary function testing. However, current evidence is primarily derived from exploratory studies, and heterogeneity exists across studies in terms of population characteristics and methodologies. This highlights the need for further standardized research to improve the consistency and reproducibility of findings.
References
1. Shen HH, Zhang M, Huang KW. Guidelines for the prevention and management of bronchial asthma (2024 edition). Zhonghua Jie He He Hu Xi Za Zhi 2025;48:208-48.
2. GBD 2021 Causes of Death Collaborators. Global burden of 288 causes of death and life expectancy decomposition in 204 countries and territories and 811 subnational locations, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet 2024;403:2100-32.
3. Rattray NJ, Hamrang Z, Trivedi DK, Goodacre R, Fowler SJ. Taking your breath away: metabolomics breathes life into personalized medicine. Trends Biotechnol 2014;32:538-48.
4. Yu KL, Yang HC, Lee CF, Wu SY, Ye ZK, Rai SK, et al. Exhaled breath analysis using a novel electronic nose for different respiratory disease entities. Lung 2025;203:14.
5. Haick H, Broza YY, Mochalski P, Ruzsanyi V, Amann A. Assessment, origin, and implementation of breath volatile cancer markers. Chem Soc Rev 2014;43:1423-49.
6. Behera B, Joshi R, Anil Vishnu GK, Bhalerao S, Pandya HJ. Electronic nose: a non-invasive technology for breath analysis of diabetes and lung cancer patients. J Breath Res 2019;13:024001.
7. Chen H, Huo D, Zhang J. Gas recognition in e-nose system: a review. IEEE Trans Biomed Circuits Syst 2022;16:169-84.
8. Wilson AD, Baietto M. Advances in electronic-nose technologies developed for biomedical applications. Sensors (Basel) 2011;11:1105-76.
9. Röck F, Barsan N, Weimar U. Electronic nose: current status and future trends. Chem Rev 2008;108:705-25.
10. Töreyin ZN, Ghosh M, Göksel Ö, Göksel T, Godderis L. Exhaled breath analysis in diagnosis of malignant pleural mesothelioma: systematic review. Int J Environ Res Public Health 2020;17:1110.
11. Dragonieri S, Pennazza G, Carratu P, Resta O. Electronic nose technology in respiratory diseases. Lung 2017;195:157-65.
12. Li Y, Wei X, Zhou Y, Wang J, You R. Research progress of electronic nose technology in exhaled breath disease analysis. Microsyst Nanoeng 2023;9:129.
13. Farraia MV, Cavaleiro Rufo J, Paciência I, Mendes F, Delgado L, Moreira A. The electronic nose technology in clinical diagnosis: a systematic review. Porto Biomed J 2019;4:e42.
14. Bannier M, van de Kant KDG, Jöbsis Q, Dompeling E. Feasibility and diagnostic accuracy of an electronic nose in children with asthma and cystic fibrosis. J Breath Res 2019;13:036009.
15. van der Sar IG, Wijsenbeek MS, Braunstahl GJ, Loekabino JO, Dingemans AC, In 't Veen J, et al. Differentiating interstitial lung diseases from other respiratory diseases using electronic nose technology. Respir Res 2023;24:271.
16. Tirzīte M, Bukovskis M, Strazda G, Jurka N, Taivans I. Detection of lung cancer in exhaled breath with an electronic nose using support vector machine analysis. J Breath Res 2017;11:036009.
17. Rutjes N, Van den Bongaardt I, Hashimoto S, Sterk P, Van Aalderen W, Terheggen-Lagro S, et al. Prediction of asthma in early preschool wheezing by electronic nose analysis. Pediatr Allergy Immunol 2022;33:e13612.
18. Farraia M, Cavaleiro Rufo J, Paciência I, Castro Mendes F, Rodolfo A, Rama T, et al. Human volatilome analysis using eNose to assess uncontrolled asthma in a clinical setting. Allergy 2020;75:1630-9.
19. Tenero L, Sandri M, Piazza M, Paiola G, Zaffanello M, Piacentini G. Electronic nose in discrimination of children with uncontrolled asthma. J Breath Res 2020;14:046003.
20. Fens N, van der Schee MP, Brinkman P, Sterk PJ. Exhaled breath analysis by electronic nose in airways disease. Established issues and key questions. Clin Exp Allergy 2013;43:705-15.
21. de Vries R, Dagelet YWF, Spoor P, Snoey E, Jak PMC, Brinkman P, et al. Clinical and inflammatory phenotyping by breathomics in chronic airway diseases irrespective of the diagnostic label. Eur Respir J 2018;51:1701817.
22. Abdel-Aziz MI, Brinkman P, Vijverberg SJH, Neerincx AH, de Vries R, Dagelet YWF, et al. eNose breath prints as a surrogate biomarker for classifying patients with asthma by atopy. J Allergy Clin Immunol 2020;146:1045-55.
23. Brinkman P, Wagener AH, Hekking PP, Bansal AT, Maitland-van der Zee AH, Wang Y, et al. Identification and prospective stability of electronic nose (eNose)-derived inflammatory phenotypes in patients with severe asthma. J Allergy Clin Immunol 2019;143:1811-20.e7.
24. van der Schee MP, Palmay R, Cowan JO, Taylor DR. Predicting steroid responsiveness in patients with asthma using exhaled breath profiling. Clin Exp Allergy 2013;43:1217-25.
25. Brinkman P, Ahmed WM, Gómez C, Knobel HH, Weda H, Vink TJ, et al. Exhaled volatile organic compounds as markers for medication use in asthma. Eur Respir J 2020;55:1900544.
26. Gaugg MT, Engler A, Nussbaumer-Ochsner Y, Bregy L, Stöberl AS, Gaisl T, et al. Metabolic effects of inhaled salbutamol determined by exhaled breath analysis. J Breath Res 2017;11:046004.
27. Zeng J, Usemann J, Singh KD, Jochmann A, Trachsel D, Frey U, et al. Pharmacometabolomics via real-time breath analysis captures metabotypes of asthmatic children associated with salbutamol responsiveness. iScience 2024;27:111446.
28. Brinkman P, van de Pol MA, Gerritsen MG, Bos LD, Dekker T, Smids BS, et al. Exhaled breath profiles in the monitoring of loss of control and clinical recovery in asthma. Clin Exp Allergy 2017;47:1159-69.
29. Buszewski B, Ulanowska A, Ligor T, Denderz N, Amann A. Analysis of exhaled breath from smokers, passive smokers and non-smokers by solid-phase microextraction gas chromatography/mass spectrometry. Biomed Chromatogr 2009;23:551-6.
30. Gaida A, Holz O, Nell C, Schuchardt S, Lavae-Mokhtari B, Kruse L, et al. A dual center study to compare breath volatile organic compounds from smokers and non-smokers with and without COPD. J Breath Res 2016;10:026006.
31. Filipiak W, Ruzsanyi V, Mochalski P, Filipiak A, Bajtarevic A, Ager C, et al. Dependence of exhaled breath composition on exogenous factors, smoking habits and exposure to air pollutants. J Breath Res 2012;6:036008.
32. Blanchet L, Smolinska A, Baranska A, Tigchelaar E, Swertz M, Zhernakova A, et al. Factors that influence the volatile organic compound content in human breath. J Breath Res 2017;11:016013.
33. Cheng ZJ, Warwick G, Yates DH, Thomas PS. An electronic nose in the discrimination of breath from smokers and non-smokers: a model for toxin exposure. J Breath Res 2009;3:036003.
34. Capone S, Tufariello M, Forleo A, Longo V, Giampetruzzi L, Radogna AV, et al. Chromatographic analysis of VOC patterns in exhaled breath from smokers and nonsmokers. Biomed Chromatogr 2018;32:e4132.
35. Chen X, Wang F, Lin L, Dong H, Huang F, Ghulam Muhammad K, et al. Association of smoking with metabolic volatile organic compounds in exhaled breath. Int J Mol Sci 2017;18:2235.
36. Adcock IM, Caramori G, Barnes PJ. Chronic obstructive pulmonary disease and lung cancer: new molecular insights. Respiration 2011;81:265-84.
37. Seet RC, Lee CY, Loke WM, Huang SH, Huang H, Looi WF, et al. Biomarkers of oxidative damage in cigarette smokers: which biomarkers might reflect acute versus chronic oxidative stress? Free Radic Biol Med 2011;50:1787-93.
38. Principe S, van Bragt J, Longo C, de Vries R, Sterk PJ, Scichilone N, et al. The influence of smoking status on exhaled breath profiles in asthma and COPD patients. Molecules 2021;26:1357.
39. Bikov A, Lazar Z, Schandl K, Antus BM, Losonczy G, Horvath I. Exercise changes volatiles in exhaled breath assessed by an electronic nose. Acta Physiol Hung 2011;98:321-8.
40. Bikov A, Galffy G, Tamasi L, Bartusek D, Antus B, Losonczy G, et al. Exhaled breath condensate pH decreases during exercise-induced bronchoconstriction. Respirology 2014;19:563-9.
41. Dragonieri S, Marco MD, Ahroud M, Quaranta VN, Portacci A, Iorillo I, et al. Electronic nose based analysis of exhaled volatile organic compounds spectrum reveals asthmatic shifts and consistency in controls post-exercise and spirometry. J Breath Res 2024;18:036006.
42. Horváth I, Hunt J, Barnes PJ, Alving K, Antczak A, Baraldi E, et al. Exhaled breath condensate: methodological recommendations and unresolved questions. Eur Respir J 2005;26:523-48.
43. Amann A, Costello Bde L, Miekisch W, Schubert J, Buszewski B, Pleil J, et al. The human volatilome: volatile organic compounds (VOCs) in exhaled breath, skin emanations, urine, feces and saliva. J Breath Res 2014;8:034001.
44. Miekisch W, Schubert JK, Noeldge-Schomburg GF. Diagnostic potential of breath analysis--focus on volatile organic compounds. Clin Chim Acta 2004;347:25-39.
45. Oyerinde AS, Selvaraju V, Babu JR, Geetha T. Potential role of oxidative stress in the production of volatile organic compounds in obesity. Antioxidants (Basel) 2023;12:129.
46. Fomčenko I, Bikulčienė I, Karčiauskaitė D, Urbonas M, Alekna V, Šapoka V. Age-related variations in body composition and metabolic health: a cross-sectional study in adults. Medicina (Kaunas) 2025;61:1951.
47. Stanojevic S, Kaminsky DA, Miller MR, Thompson B, Aliverti A, Barjaktarevic I, et al. ERS/ATS technical standard on interpretive strategies for routine lung function tests. Eur Respir J 2022;60:2101499.
48. Reddel HK, Bacharier LB, Bateman ED, Brightling CE, Brusselle GG, Buhl R, et al. Global initiative for asthma strategy 2021: executive summary and rationale for key changes. Am J Respir Crit Care Med 2022;205:17-35.
49. Greiter MB, Keck L, Siegmund T, Hoeschen C, Oeh U, Paretzke HG. Differences in exhaled gas profiles between patients with type 2 diabetes and healthy controls. Diabetes Technol Ther 2010;12:455-63.
50. Minh TD, Oliver SR, Ngo J, Flores R, Midyett J, Meinardi S, et al. Noninvasive measurement of plasma glucose from exhaled breath in healthy and type 1 diabetic subjects. Am J Physiol Endocrinol Metab 2011;300:E1166-75.
51. Chan MJ, Li YJ, Wu CC, Lee YC, Zan HW, Meng HF, et al. Breath ammonia is a useful biomarker predicting kidney function in chronic kidney disease patients. Biomedicines 2020;8:468.
52. Waltman CG, Marcelissen TAT, van Roermund JGH. Exhaled-breath testing for prostate cancer based on volatile organic compound profiling using an electronic nose device (Aeonose™): a preliminary report. Eur Urol Focus 2020;6:1220-5.
53. Phillips M, Basa-Dalay V, Bothamley G, Cataneo RN, Lam PK, Natividad MP, et al. Breath biomarkers of active pulmonary tuberculosis. Tuberculosis (Edinb) 2010;90:145-51.
54. Timms C, Thomas PS, Yates DH. Detection of gastro-oesophageal reflux disease (GORD) in patients with obstructive lung disease using exhaled breath profiling. J Breath Res 2012;6:016003.
55. Fens N, Zwinderman AH, van der Schee MP, de Nijs SB, Dijkers E, Roldaan AC, et al. Exhaled breath profiling enables discrimination of chronic obstructive pulmonary disease and asthma. Am J Respir Crit Care Med 2009;180:1076-82.
56. de Vries R, Brinkman P, van der Schee MP, Fens N, Dijkers E, Bootsma SK, et al. Integration of electronic nose technology with spirometry: validation of a new approach for exhaled breath analysis. J Breath Res 2015;9:046001.
57. Boots AW, van Berkel JJ, Dallinga JW, Smolinska A, Wouters EF, van Schooten FJ. The versatile use of exhaled volatile organic compounds in human health and disease. J Breath Res 2012;6:027108.
58. Miekisch W, Kischkel S, Sawacki A, Liebau T, Mieth M, Schubert JK. Impact of sampling procedures on the results of breath analysis. J Breath Res 2008;2:026007.
59. van de Kant KD, van der Sande LJ, Jöbsis Q, van Schayck OC, Dompeling E. Clinical use of exhaled volatile organic compounds in pulmonary diseases: a systematic review. Respir Res 2012;13:117.
60. Montuschi P, Mores N, Trové A, Mondino C, Barnes PJ. The electronic nose in respiratory medicine. Respiration 2013;85:72-84.
61. van der Schee MP, Paff T, Brinkman P, van Aalderen WMC, Haarman EG, Sterk PJ. Breathomics in lung disease. Chest 2015;147:224-31.
Downloads
Published
Issue
Section
License
Copyright (c) 2026 The Author(s)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Mattioli 1885 has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.




